The Halogens
Before we can learn about Fluorine, we have to learn about the family it comes from!
The word "halogen" means salt forming and comes from "halos" which means salt in Greek. The halogens are found on the periodic table of elements at group 17. Group 17 consists of chlorine, bromine, iodine, and fluorine. The halogens are used for things such as disinfectants, purifiers, and fumigants. In other words, the halogens are used to better the qualities of other compounds. For example, fluorine is added to city tap water to disinfect the water. Also, chlorine is used to purify many substances from plastic to polymers. The halogens are all non-metal and have seven electrons in their outer shell.
Where the halogens are located on the periodic table. (Highlighted in green)
The Halogens, in terms of electrons!
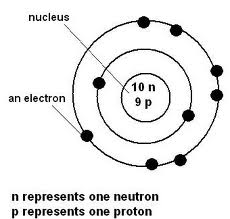
The halogens all have seven electrons in their outer shell. These electrons are known as "valance electrons" The innermost energy level(the ring with two circles) holds up to two electrons, and the second and third energy levels hold eight electrons. In the halogen's case, they always have seven valance electrons.
You can navigate the website through the tabs on the top of the website!